News
News from IEMC
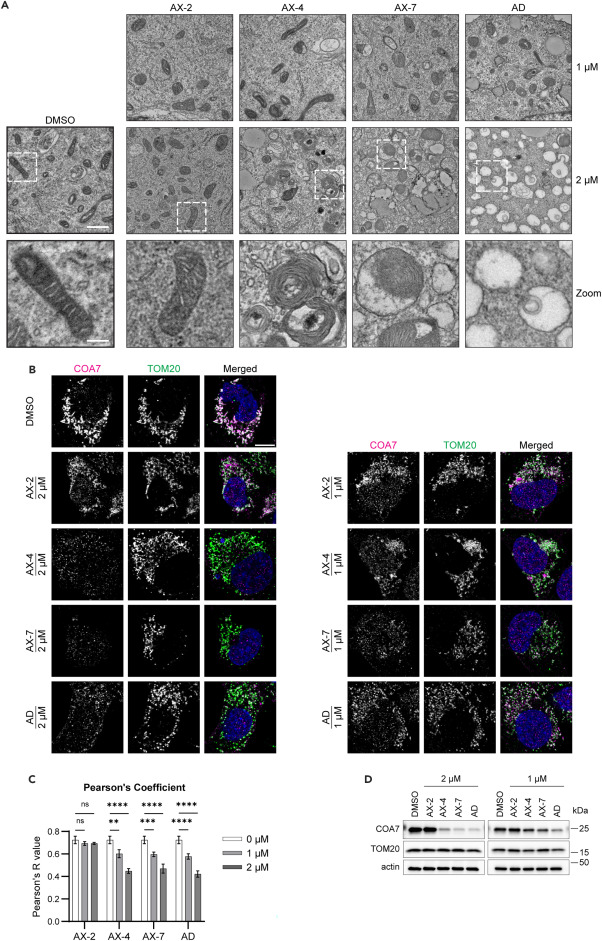
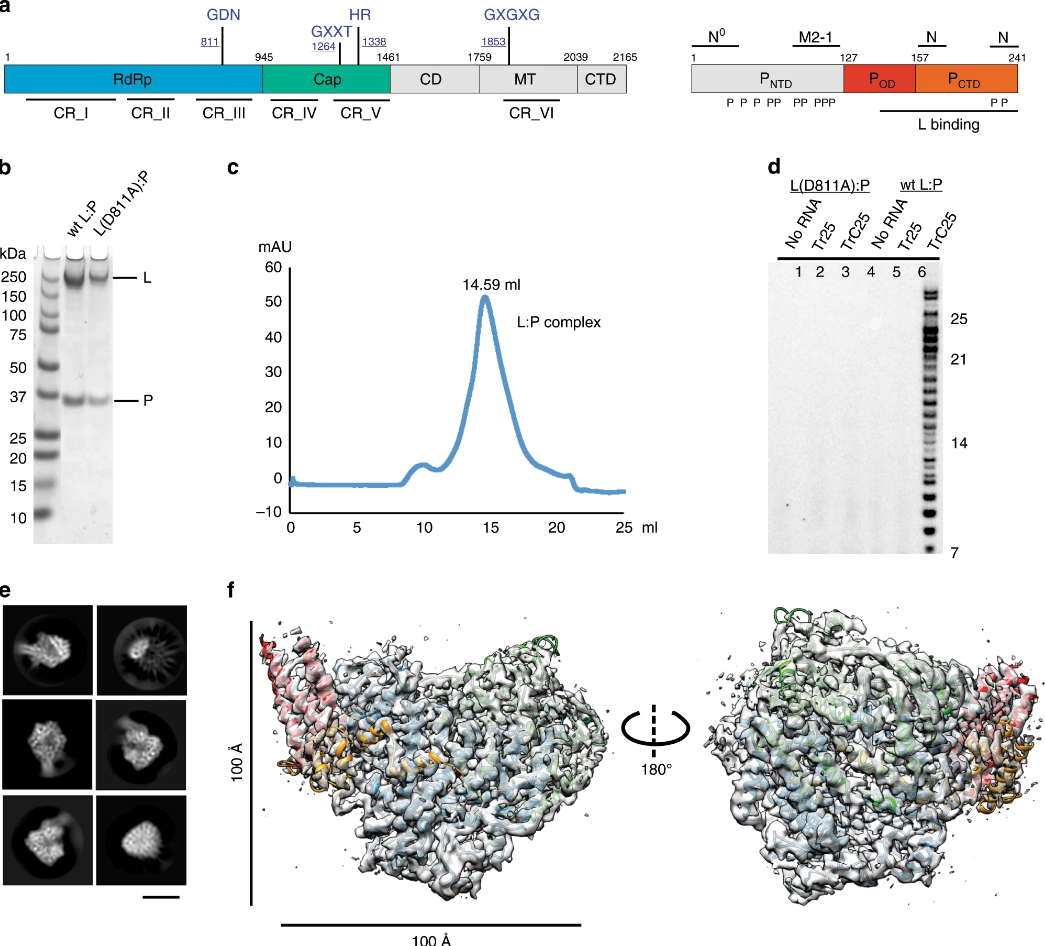
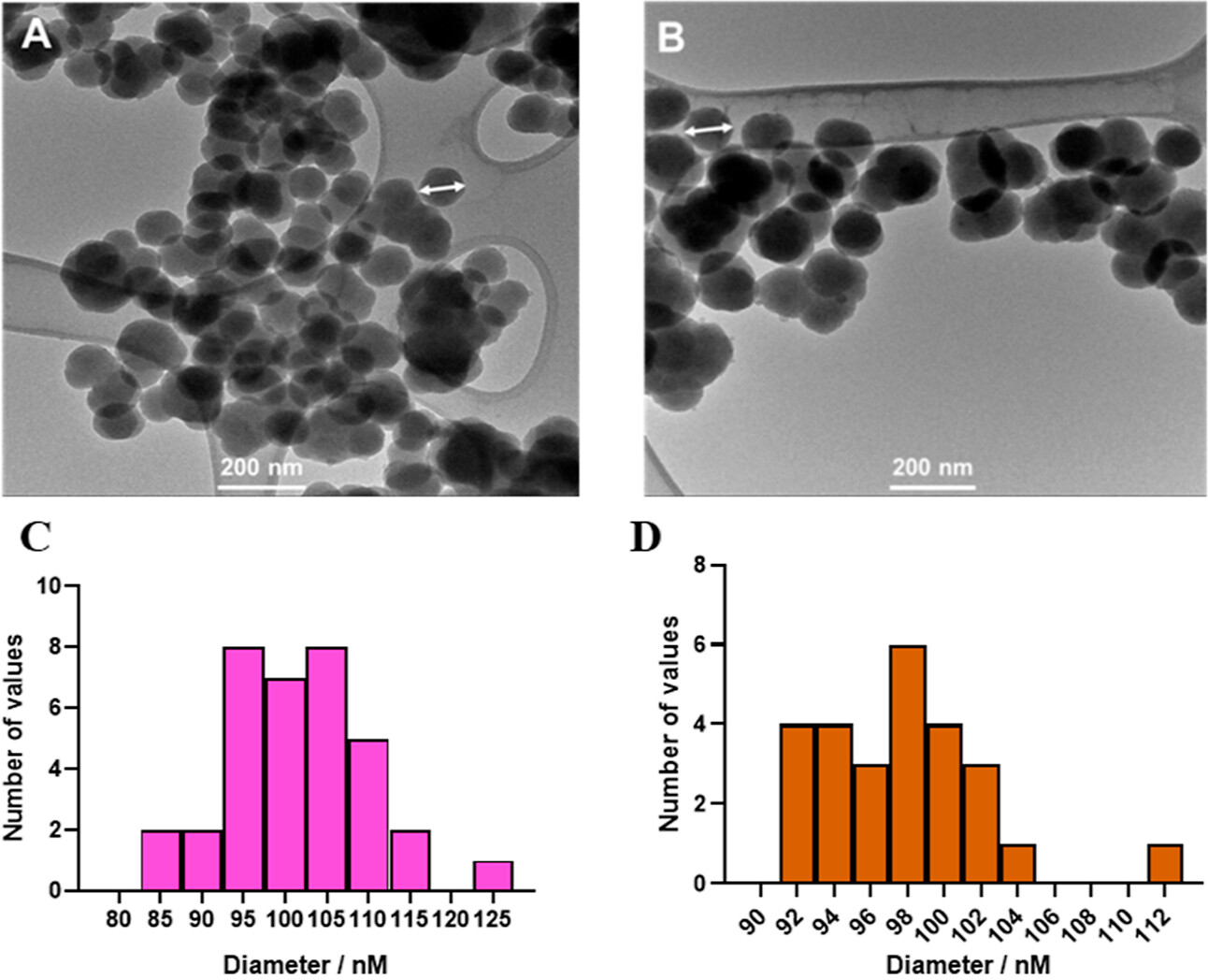
Electron micrographs used for these figures were acquired at the IEMC. Thank you for acknowledging our contribution.
IEMC News
Our ThermoFisher (TFS) Hydra Bio Plasma Focused Ion Beam – Scanning Electron Microscope (pFIB-SEM) is ready and available to researchers. The pFIB-SEM technology allows biomedical investigators in human health and human diseases to understand the three-dimensional organization of cellular environments and have access to state-of-the-art research techniques. This instrument is also equipped with a Delmic Meteor integrated Fluorescent Light Microscope (iFLM), which allows for Correlated Light and Electron Microscopy (CLEM) methods.
With this new technology, the IEMC will support structural research of cells and macromolecular complexes involved in infectious diseases, neurodegenerative disorders, heart and liver conditions, and cancer. Acquisition of this pFIB-SEM equipment with cryo-capabilities has immediate and profound impact by supporting a number of NIH-sponsored investigators at Emory University and members of the Georgia Core Facilities Partnership, who enjoy shared-access to each other’s state-of-the-art core facilities.
Electron Microscopy Events
Microscopy and Microanalysis - 2026
The meeting of the Microscopy Society of America will take place at the Baird Center in Milwaukee, WI from August 2-6, 2026. Link
SouthEastern Microscopy Society meeting - 2026
SEMS 61th annual meeting will be held May 11- 13, 2026 at the University of Georgia Center for Continuing Education and Hotel. Link